
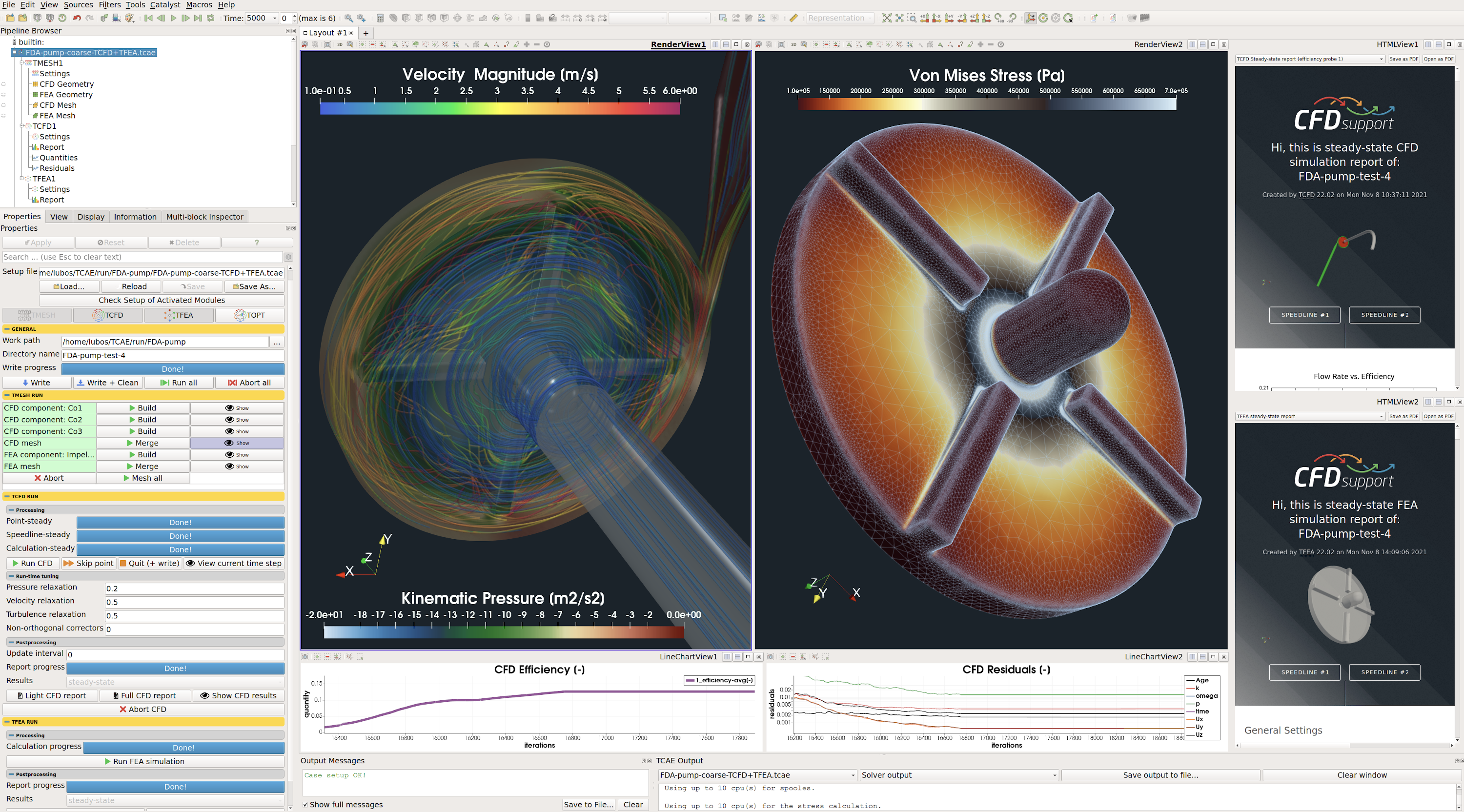
FDA Pump CFD & FEA Simulation Benchmark
This benchmark project shows a complex CFD and FEA analysis of FDA pump using simulation environment TCAE
Introduction
This study shows a complex step-by-step benchmark validation of an FDA Pump simulated in CFD & FEA simulation software, including FSI and modal analysis. The simulation software used for this analysis is TCAE – a comprehensive simulation environment based on open-source. This FDA Pump benchmark is a special U.S. Food and Drug Administration (FDA) initiative to validate CFD results for medical devices. This FDA pump was extensively tested, measured & simulated in multiple laboratories to provide experimental velocities, pressures, and hemolysis data to support CFD validation. The goal of this study is to show in detail how to make a comprehensive simulation analysis of the centrifugal blood pump characteristics: efficiency, flow coefficient, torque, power, pressure, stress, displacement, modal analysis, hemolysis, and many more. The results are compared with the experimental data [1],[2],[3].
FDA Pump - Geometry Description
The FDA pump geometry is described in the following sketch:
![FDA-pump-geometry-front-z-view-dimensions[1] FDA-pump-geometry-front-z-view-dimensions[1]](https://www.cfdsupport.com/wp-content/uploads/elementor/thumbs/FDA-pump-geometry-front-z-view-dimensions1-pjzdwbddx9u0y6nxj94waklxwbq7aqg65clgq9ybk0.png)
![FDA-pump-geometry-front-meridional-x-view-dimensions[1] FDA-pump-geometry-front-meridional-x-view-dimensions[1]](https://www.cfdsupport.com/wp-content/uploads/elementor/thumbs/FDA-pump-geometry-front-meridional-x-view-dimensions1-r0ezm4zmj4ew4eao7i9q9r4habp2ow5h5kpao2eqkg.png)
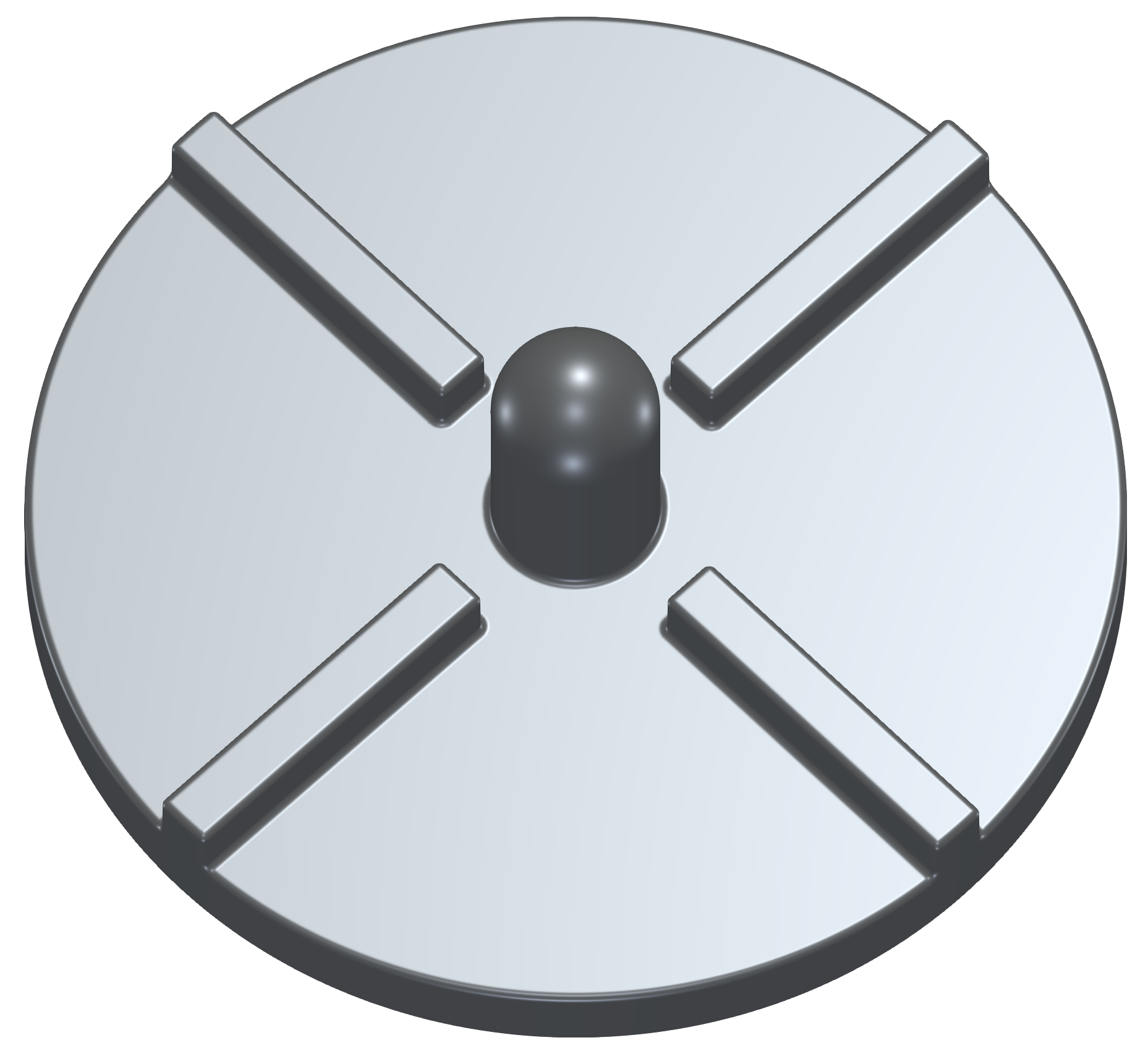
It is a small centrifugal blood pump. The acrylic rotor is a disk (5.2cm in diameter) that has four filleted blades (3mm tall and 3mm wide) orthogonally positioned on a 4mm thick rotor base, attached to a stainless steel shaft (3.2mm in diameter). A typical input for a detailed simulation analysis is a 3D surface model in form of an STL surface. For CFD simulation, it is needed to have a simulation-ready surface, which is a closed watertight model (sometimes also called waterproof, or model negative, or wet surface) of the pump inner parts where the fluid flows. For FEA simulation, it is needed to have a closed surface model of the solid of the impeller in form of a single STL surface.
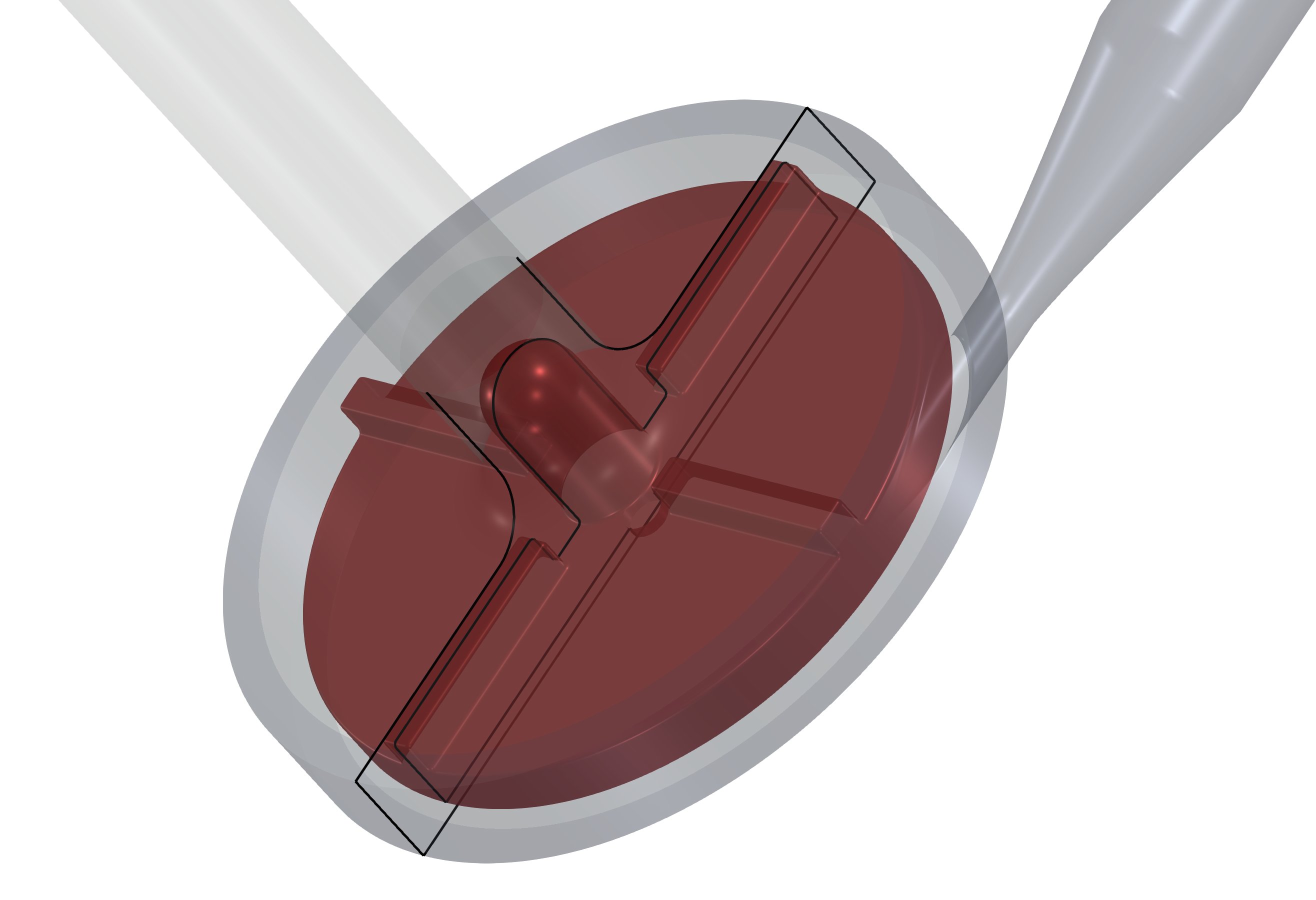
An existing CAD model of the centrifugal pump is typically received in the STEP or IGES CAD file format. Original CAD files are usually too complex for comprehensive CFD and FEA simulations, so certain preprocessing (cleaning) CAD work is generally needed. While the original CAD model for this project was simplified and cleaned using Salome open-source software, any other standard CAD system can be used instead.
FDA Pump Benchmark - CFD Preprocessing
For CFD simulation it is best to split the FDA Pump into several waterproof components because of rotation (some parts may be rotating and some parts not). Each component itself is waterproof and consists of a few or multiple STL surfaces. It is smart to split the surface model into multiple surfaces (more is better) because it opens a wider range of simulation methods (mesh refinements, manipulation, boundary conditions, evaluation of results on model parts, …).
The principle is always the same: the 3D surface model has to be created; all the tiny, irrelevant, and problematic model parts must be removed, and all the holes must be sealed up (a watertight surface model is required). This centrifugal blood pump CAD model is reasonably simple. The final surface model in the STL format is created as input for the meshing phase. This preprocessing phase of the workflow is extremely important because it determines the simulation potential and limits the CFD results.
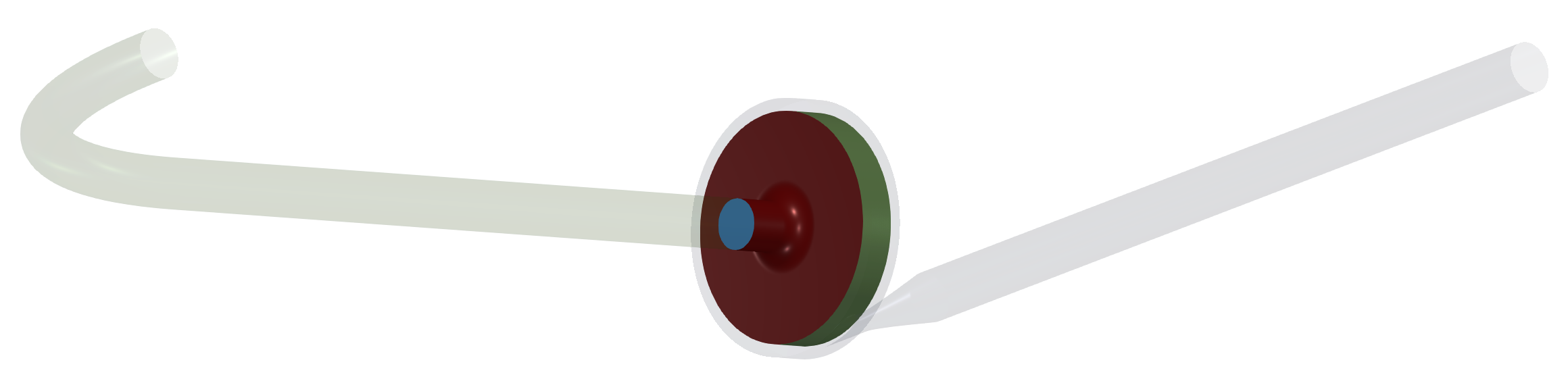
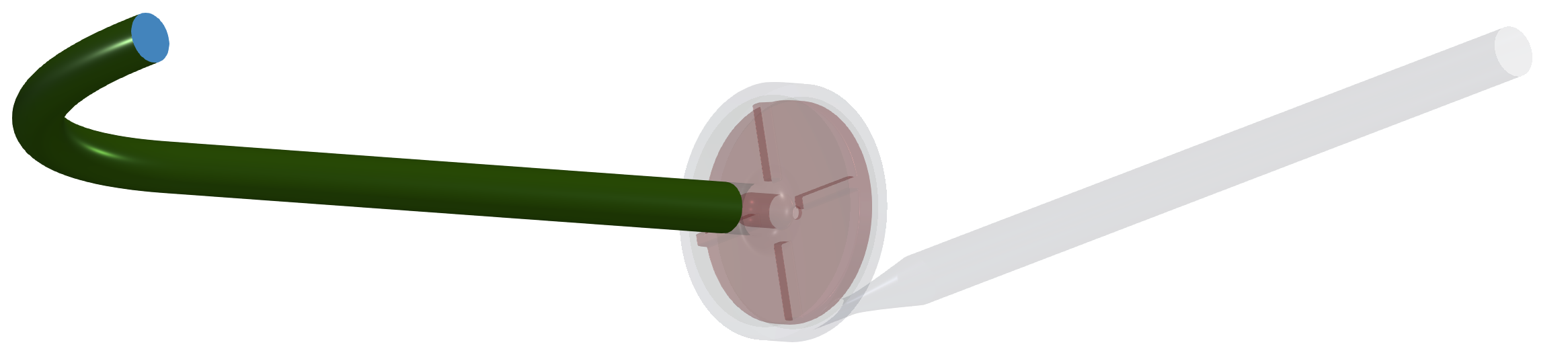
In this centrifugal pump project, the CAD model was split into three logical parts: Inlet tube, Impeller, and Volute with outlet. Each pump part (model component) is watertight and includes its own inlet interface, outlet interface, and corresponding walls (wall, blade, hub, shroud, fillets, stator blades, vane blades, …). At first, for each individual component is created its own volume mesh, and after that, in the simulation process, the component meshes are merged into a single mesh by the TCAE processor. The components are joined via interfaces. The component interfaces must fit each other topologically, while the meshes are typically different from both sides.
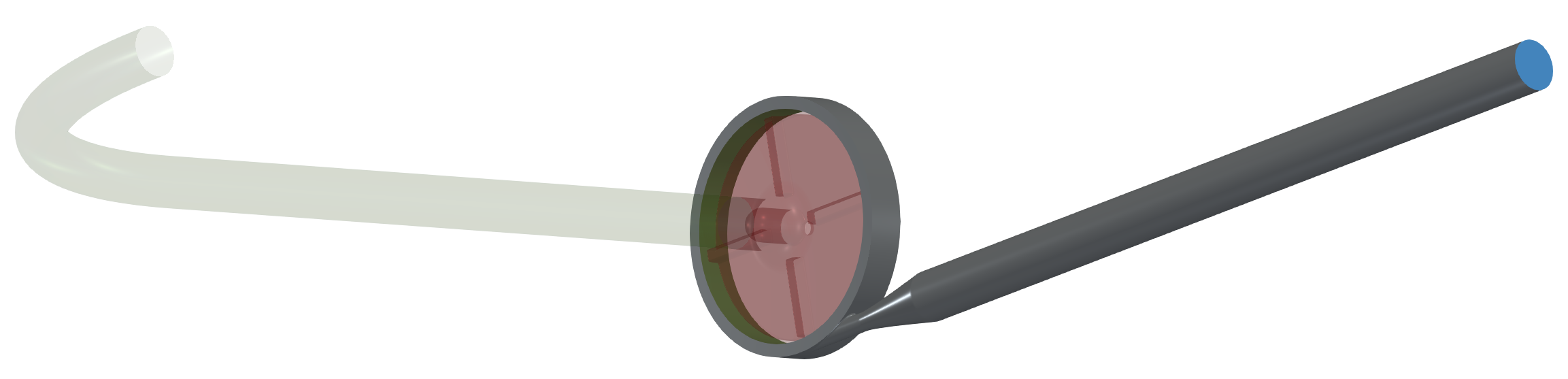
The surface model data in .stl file format together with physical inputs are loaded in TCFD. Another option would be loading an external mesh in OpenFOAM mesh format or loading an MSH mesh format (Fluent mesh format), or CGNS mesh format. This CFD methodology employs a multi-component approach, which means the model is split into a certain number of components. In TCFD each region can have its own mesh and individual meshes communicate via interfaces.
FDA Pump - FEA Preprocessing
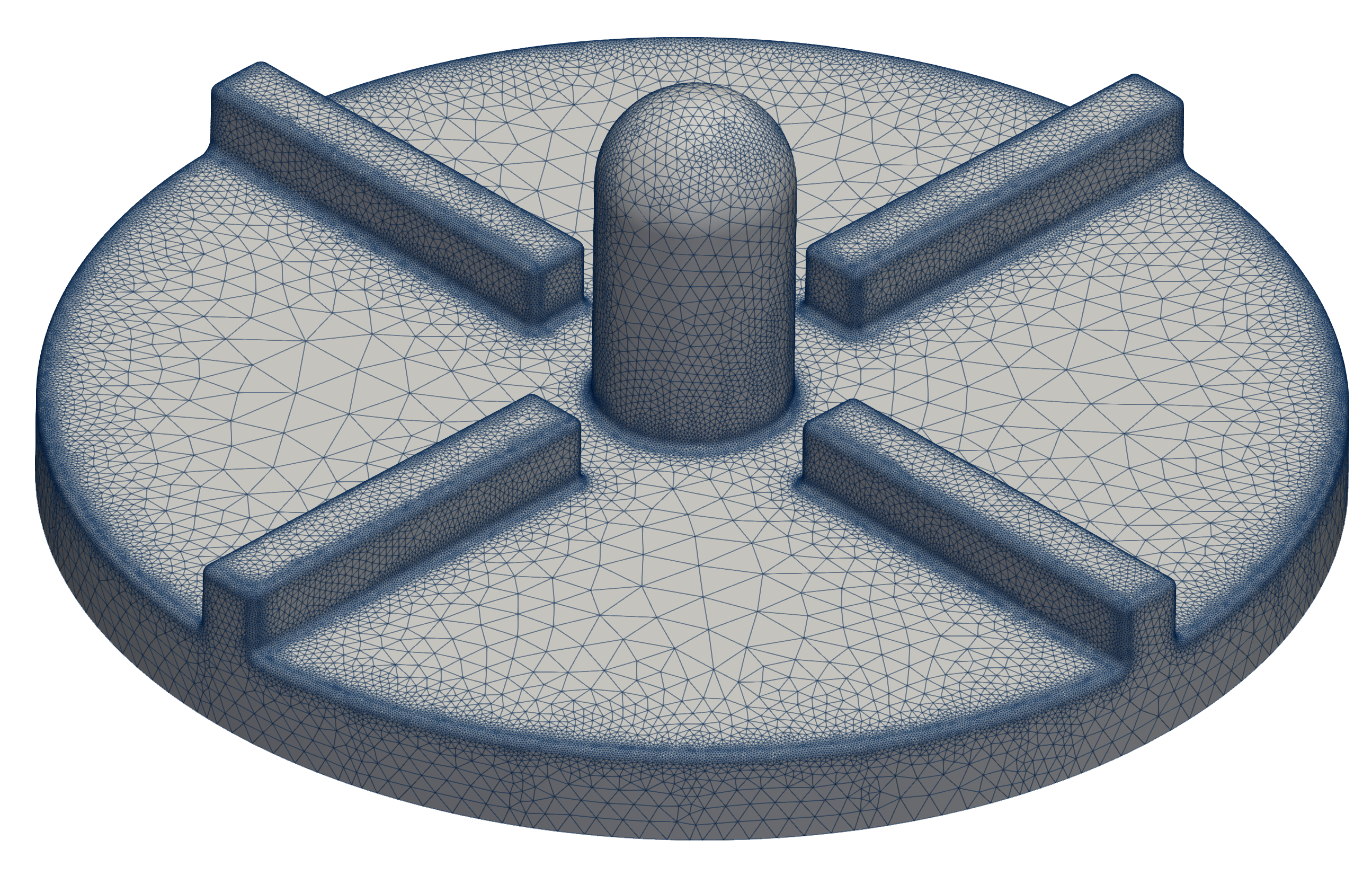
For FEA preprocessing, the rules are similar to CFD preprocessing and cleaning. It is best to create a simple, single, waterproof STL surface of the solid, for instance: rotor.stl. After that, the model is ready for meshing with the TMESH module using NetGen or Gmsh open-source application.
IMPORTANT NOTICE ON PREPROCESSING
The surface of the simulation-ready model has to be clean, simple enough but not simpler! The principles are always the same: the watertight surface model has to be created; all the tiny, irrelevant, and problematic model parts must be removed, and all the holes must be sealed up (the watertight surface model is required). The preprocessing phase is an extremely important part of each simulation workflow. It sets up all the simulation potential and limitations. It should never be underestimated. Mistakes or poor quality engineering in the preprocessing phase can be hardly compensated later in the simulation phase and postprocessing phase. For more details, see the TCAE documentation.
FDA Pump - CFD Meshing
The computational mesh for CFD is created in an automated software module TMESH, using the snappyHexMesh open-source application. All the mesh settings are done in the TCAE GUI.
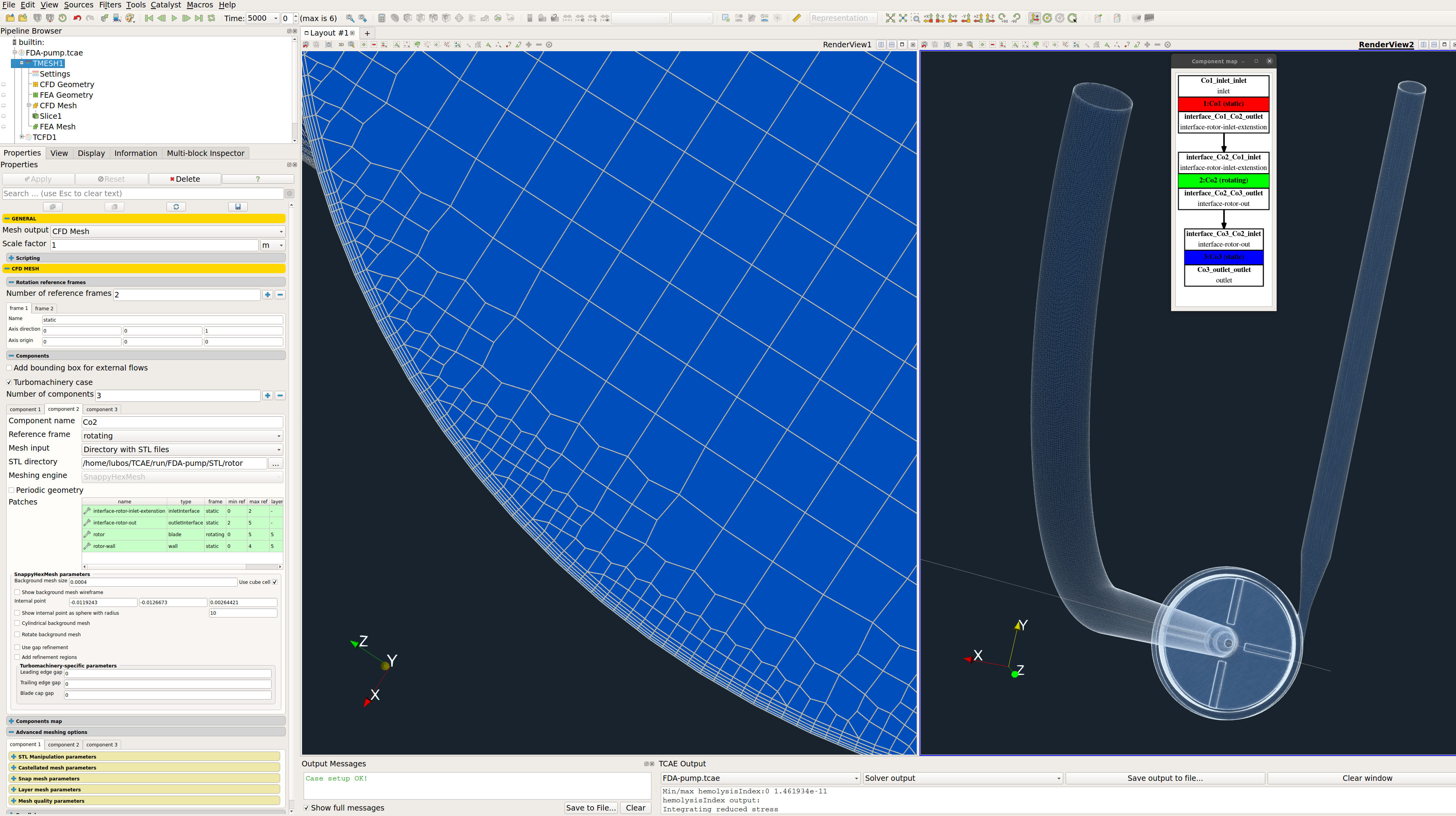
For each model component, a cartesian block mesh is created (box around the model), as an initial background mesh, that is further refined along with the simulated object. Basic mesh cell size is a cube defined with the keyword “background mesh size”.
The mesh is gradually refined to the model wall. The mesh refinement levels can be easily changed, to obtain the coarser or finer mesh, to better handle the mesh size. Inflation layers can be easily handled if needed.
FDA Pump - FEA Meshing
The computational mesh for FEA is created in an automated software module TMESH, using the NetGen open-source application. All the mesh settings are done in the TCAE GUI.
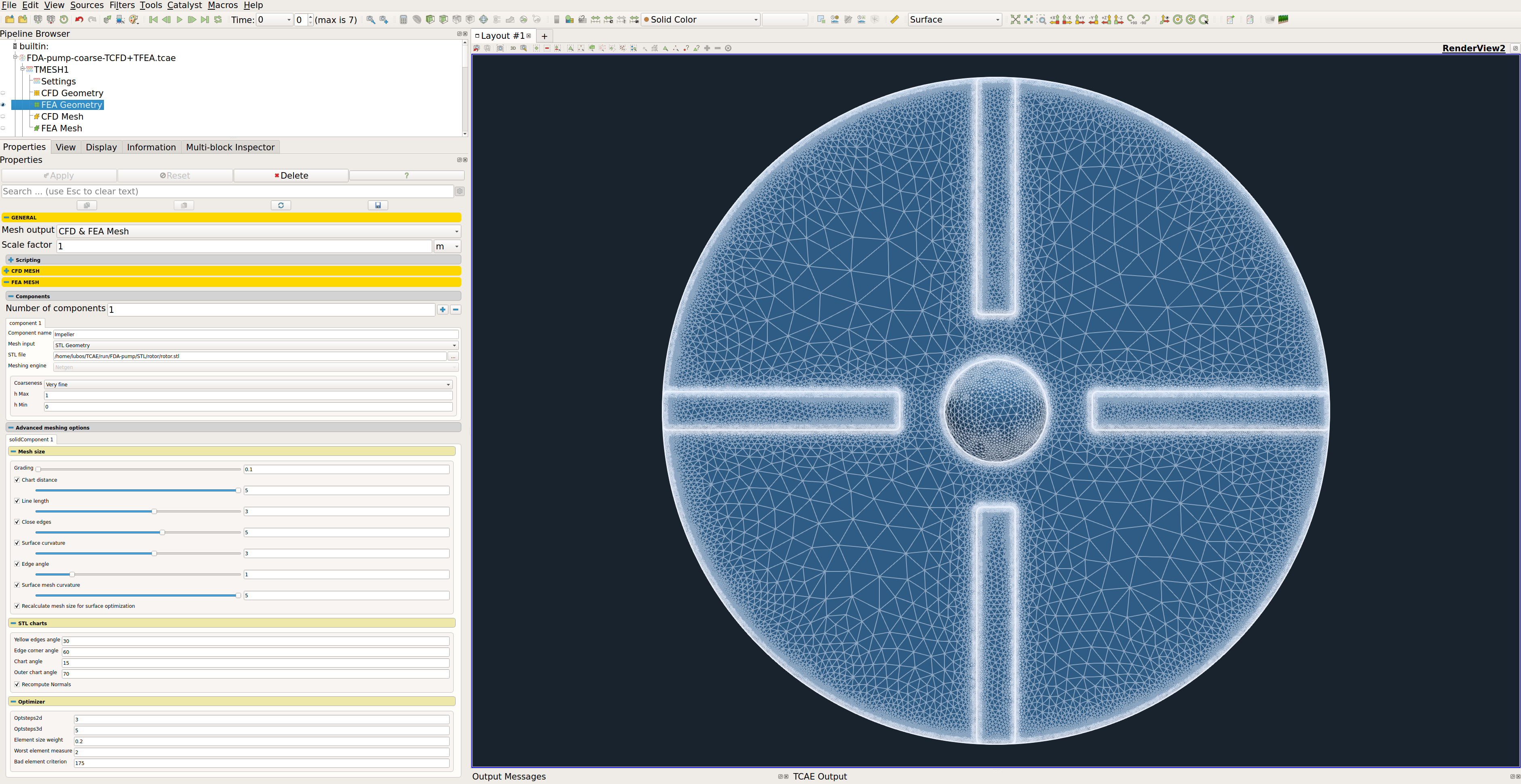
The closed STL model has been meshed with just a little effort because there are just a few parameters to set. The most important parameters for FEA meshing are “h Max” and “h Min” which mean the maximal and minimal mesh edge in meters. The mesh is created with an automated algorithm.

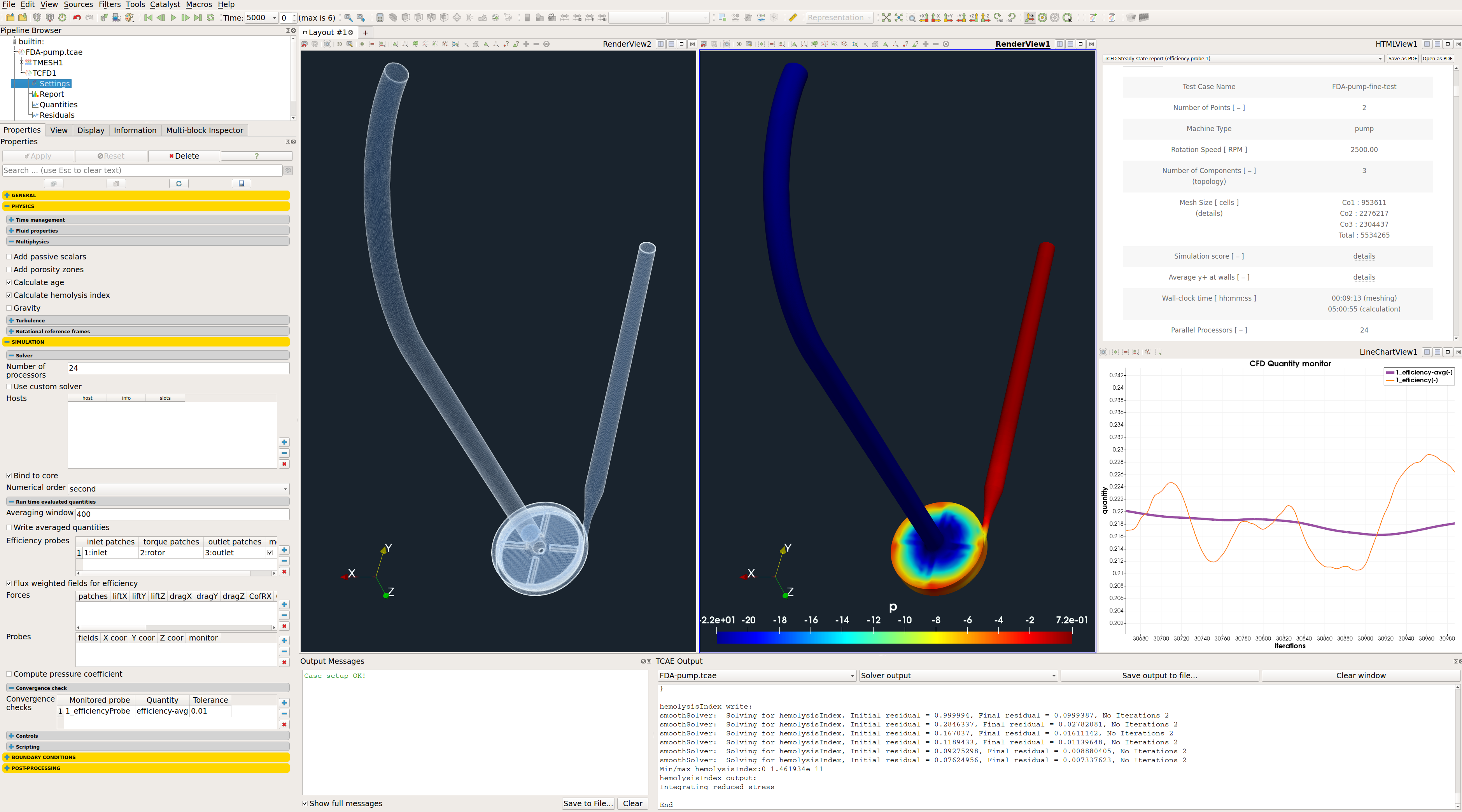
FDA Pump - CFD Simulation Setup
The CFD simulation is managed with TCAE software module TCFD. Complete CFD simulation setup and run is done in the TCFD GUI in ParaView. TCFD uses OpenFOAM open-source application.
- Simulation type: Pump
- Time management: steady-state
- Physical model: Incompressible
- Number of components: 3 [-]
- Wall roughness: none
- Speed: 2500 and 3500 [RPM]
- Outlet: Static pressure 0 [m2/s2]
- Turbulence: RANS
- Turbulence model: k-omega SST
- Shear Thinnig model: Newtonian
- Wall treatment: Wall functions
- Turbulence intensity: 5%
- Speedlines: 2 [-]
- Simulation points: 6 [-]
- Fluid: Blood
- Reference pressure: 1 [atm]
- Dynamic viscosity: 1.8 × 10E-3 [Pa⋅s]
- Water density: 1035 [kg/m3]
- CFD CPU Time: 25 core.hours/point
- BladeToBlade: on
Any project simulated in TCFD has its component graph. The component graph shows how the components are organized – the flow model topology, the inlet, the outlet, and how the components are connected via interfaces. A simple scheme of the component graph is shown below.
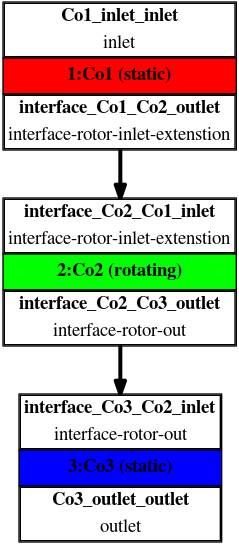
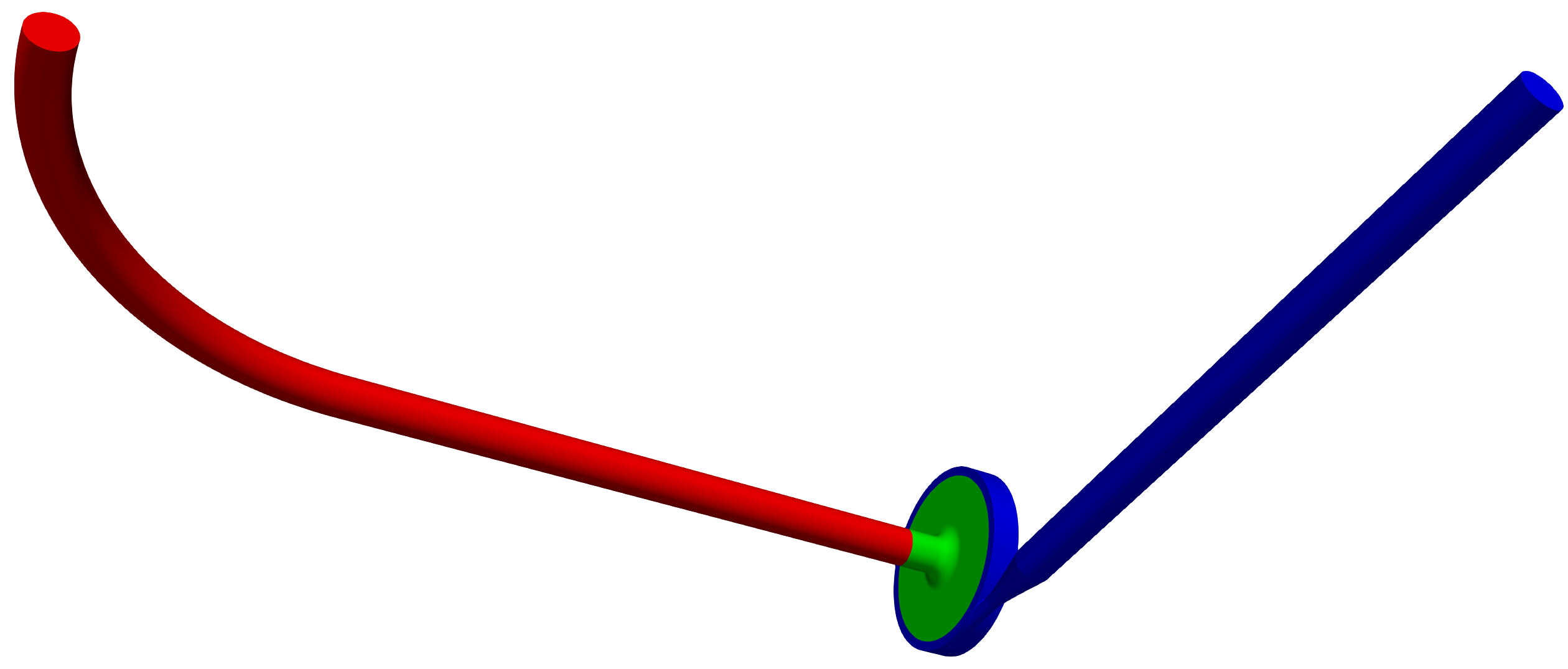
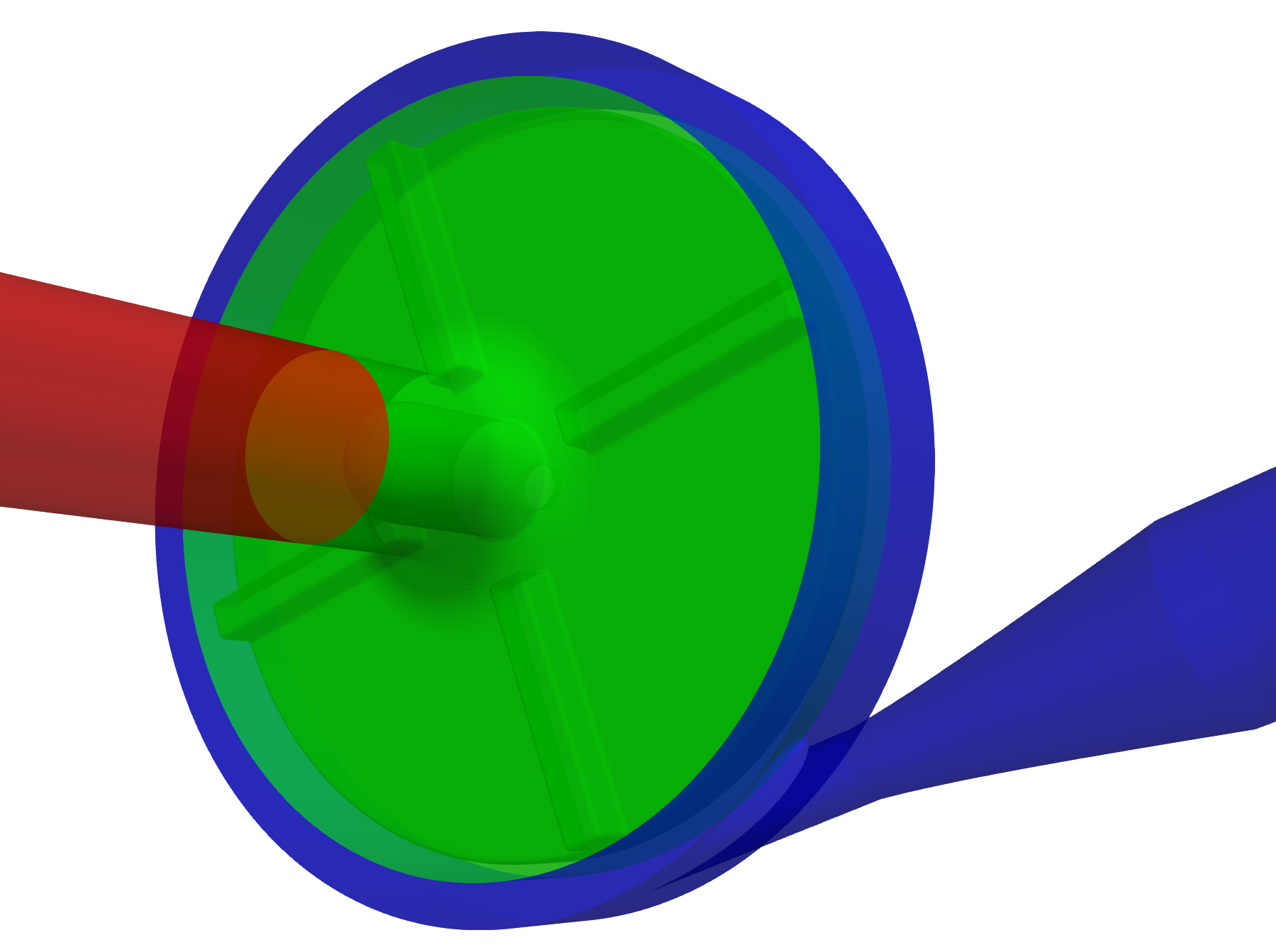
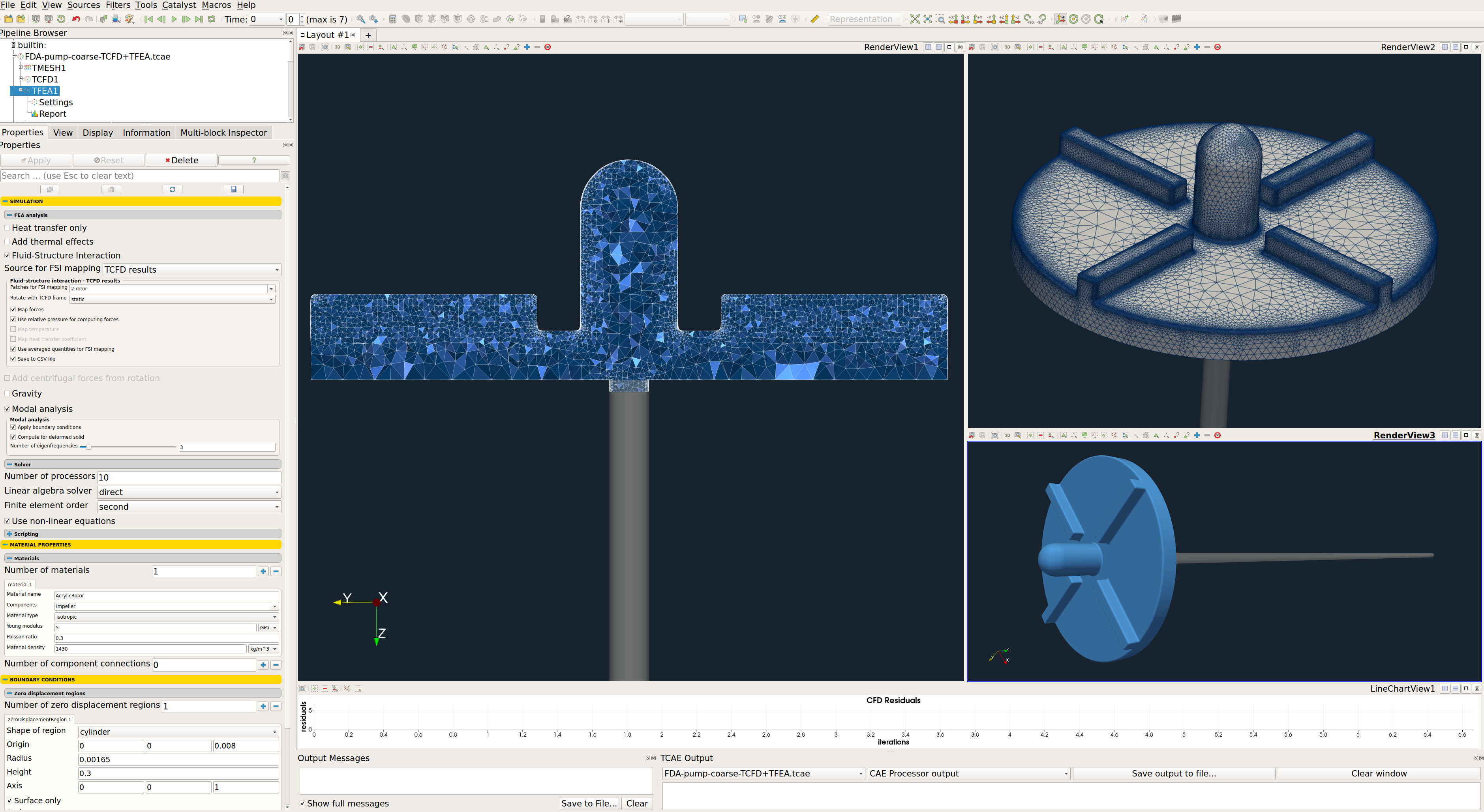
FDA Pump - FEA Simulation Setup
The FEA simulation is managed with TCAE software module TFEA. Complete FEA simulation setup and run are done in the TFEA GUI in ParaView. TFEA is based on the Calculix open-source application.
- Impeller material: Acrylic
- Material structure: isotropic
- Young modulus: 5.0E9 Pa
- Material density: 1430 kg/m3
- FSI source: TCFD [-]
- Poisson ratio: 0.3
- Fixed radius: 3.2 [mm]
- Finite element order: second
- FEA CPU Time: 0.1 core.hours/point
- Speed of rotation: 2500 and 3500 [RPM]
FDA Pump - TCAE Simulation
The TCAE simulation run is completely automated. The whole workflow can be run by a single click in the GUI, or the whole process can be run in batch mode on a background. Modules used are TCAD, TMESH, TCFD, and TFEA. The simulation is executed in the steady-state mode, for six volumetric flow rate values at two speeds as they were measured in the experiment.
![FDA-pump-table-simulation-points[1] FDA-pump-table-simulation-points[1]](https://www.cfdsupport.com/wp-content/uploads/elementor/thumbs/FDA-pump-table-simulation-points1-r0ezm18jimd99oxzfq25gx5dtnlt9prqilv1kr0mos.png)
TCFD includes a built-in post-processing module that automatically evaluates all the required quantities, such as efficiency, torque, forces, force coefficients, flow rates, pressure, velocity, and much more. All these quantities are evaluated throughout the simulation run, and all the important data is summarized in an HTML report, which can be updated anytime during the simulation, for every run.
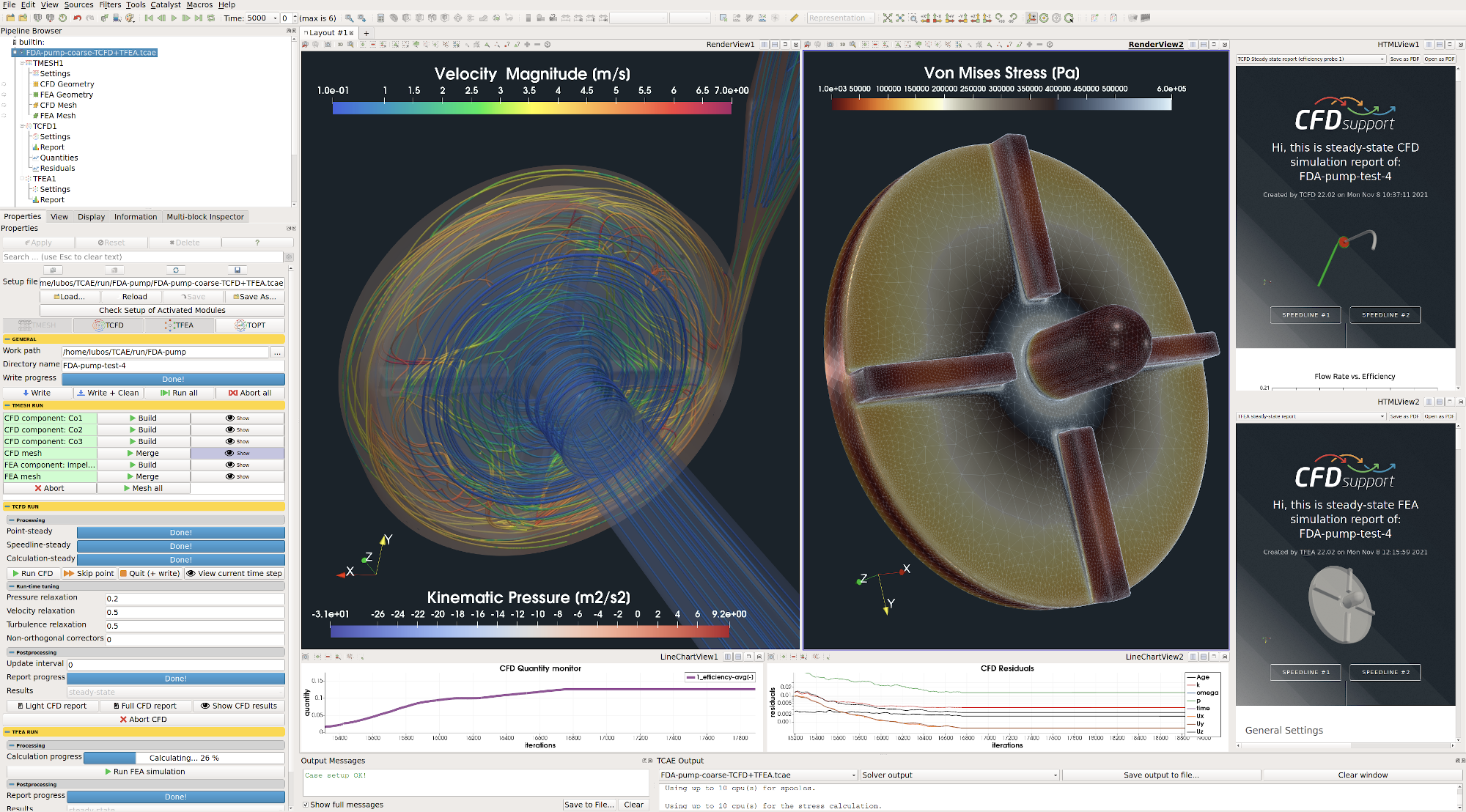
All the simulation data are also saved in tabulated .csv files for further evaluation. TCFD is capable of writing the results down at any time during the simulation. The convergence of basic quantities and integral quantities are monitored still during the simulation run. The geometry was created one-time using TCAD in the preprocessing phase. First, the TMESH is executed to create the volume meshes for CFD & FEA. Then the CFD simulation is executed and evaluated. After that, in the FSI step, the pressure field is integrated to create the force field which is prescribed as a load for the FEA simulation. Finally, the FEA simulation is executed and evaluated.
FDA Pump - Postprocessing - Integral Results
The integral results and simulation statistics are evaluated automatically for every simulation run. Every simulation run in TCAE has its own unique simulation report. The integral results both for CFD and FEA are written down in the corresponding HTML or PDF reports.
All the relevant integral quantities are evaluated, sorted, and stored in the corresponding CSV files and are ready for further usage. Following integral quantities are evaluated every time step.
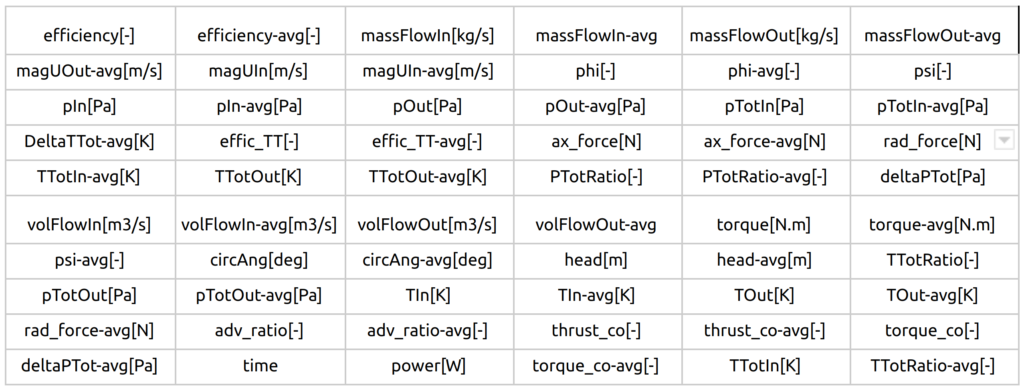
FDA Pump - Postprocessing - Volume Fields
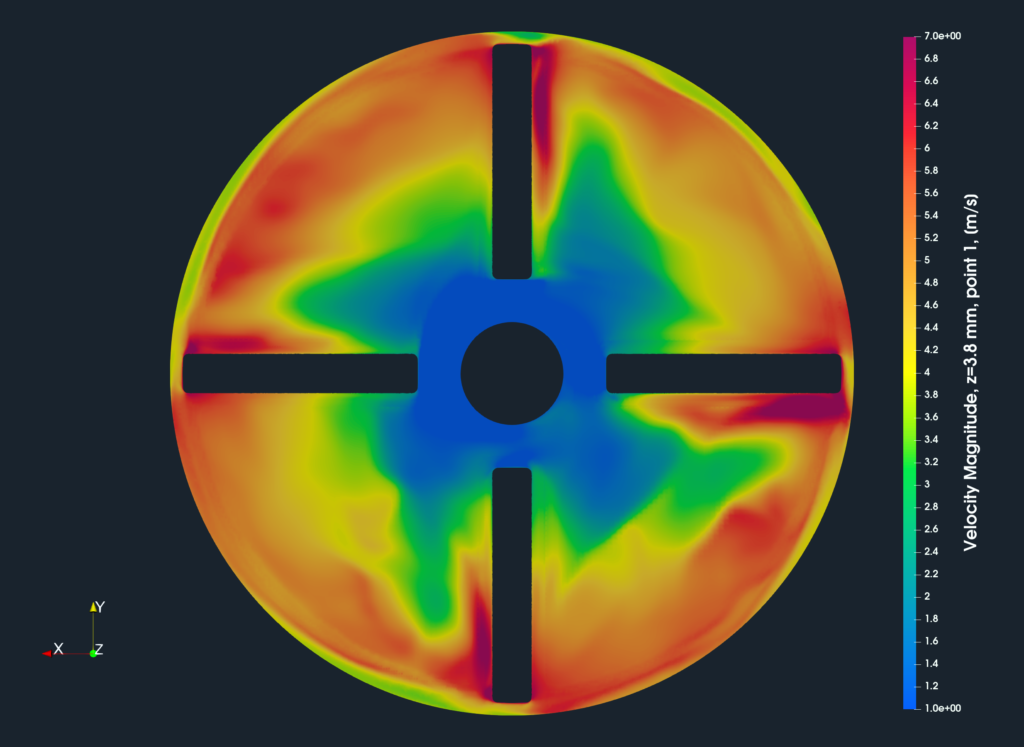
While all the integral results are stored in the CSV files and are available for further postprocessing, the volume fields are post-processed in the open-source visualization tool ParaView. ParaView provides a wide range of tools and methods for CFD & FEA postprocessing and results evaluation. There are available countless useful filters and sources, for example, Calculator, Contour, Clip, Slice, Threshold, Glyph (Vectors), Streamtraces (Streamlines), and many others.
CFD Volume Fields
FDA Pump - Postprocessing - Turbomachinery Specific Views
FDA Pump - Meridional View
For engineers who deal with rotating machinery, it is typically important to see the results, for example, total pressure or velocity, circumferentially averaged and projected on the meridian plane. This visualization method is called the Meridional Average. This meridional average projection avoids for example the pump blades and shows how the pressure (energy) or velocity is distributed along the meridian (a 2D interpretation of flow through the pump). Meridional average provides valuable information about pressure discharge distribution. Technically, the volume field data are circumferentially averaged and interpolated on the meridional plane of certain resolution. This method allows the visualization of the between the hub and shroud without the blades.
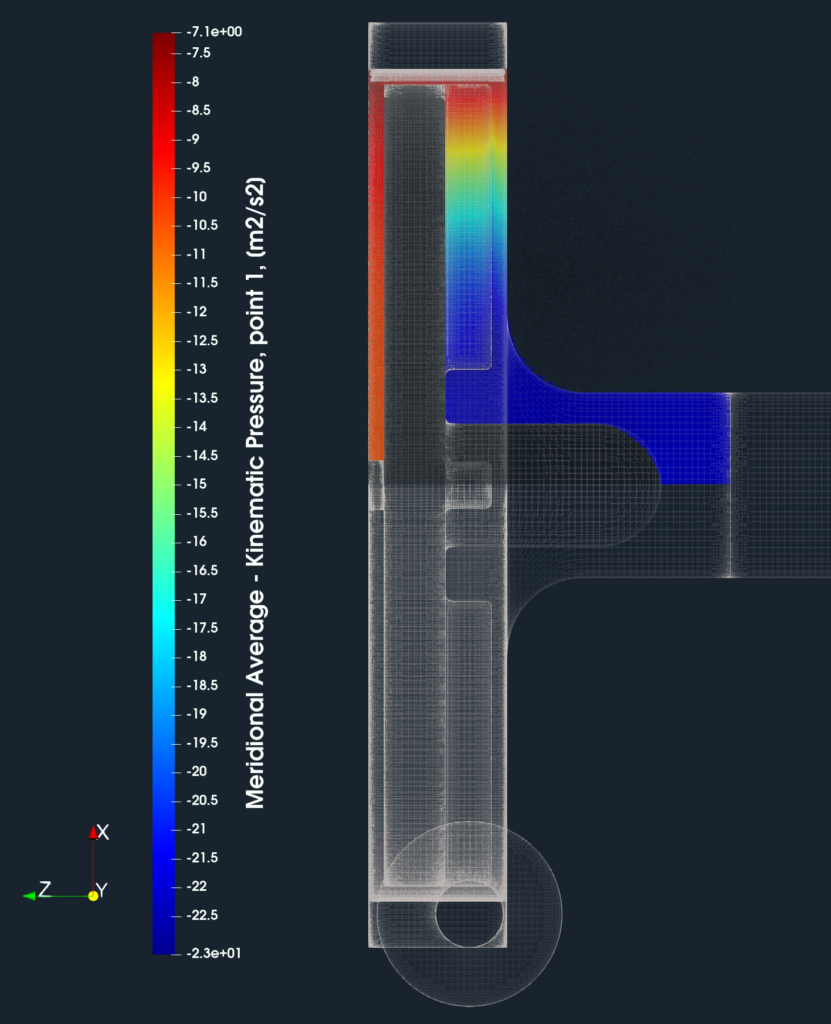
FDA Pump - Blade to Blade View
Blade to blade view is a special visualization method that transforms (unwraps) the rotational object (and its corresponding volume fields) into the dimensionless hexahedron of edges 2phi x 1 x 1. The blade-to-blade view offers a unique perspective that allows an inspection of the flow between the blades on any plane at a fixed relative distance between hub and shroud surfaces. This visualization method helps the engineers to check the angles at the leading and trailing edges at specific levels between hub and shroud.
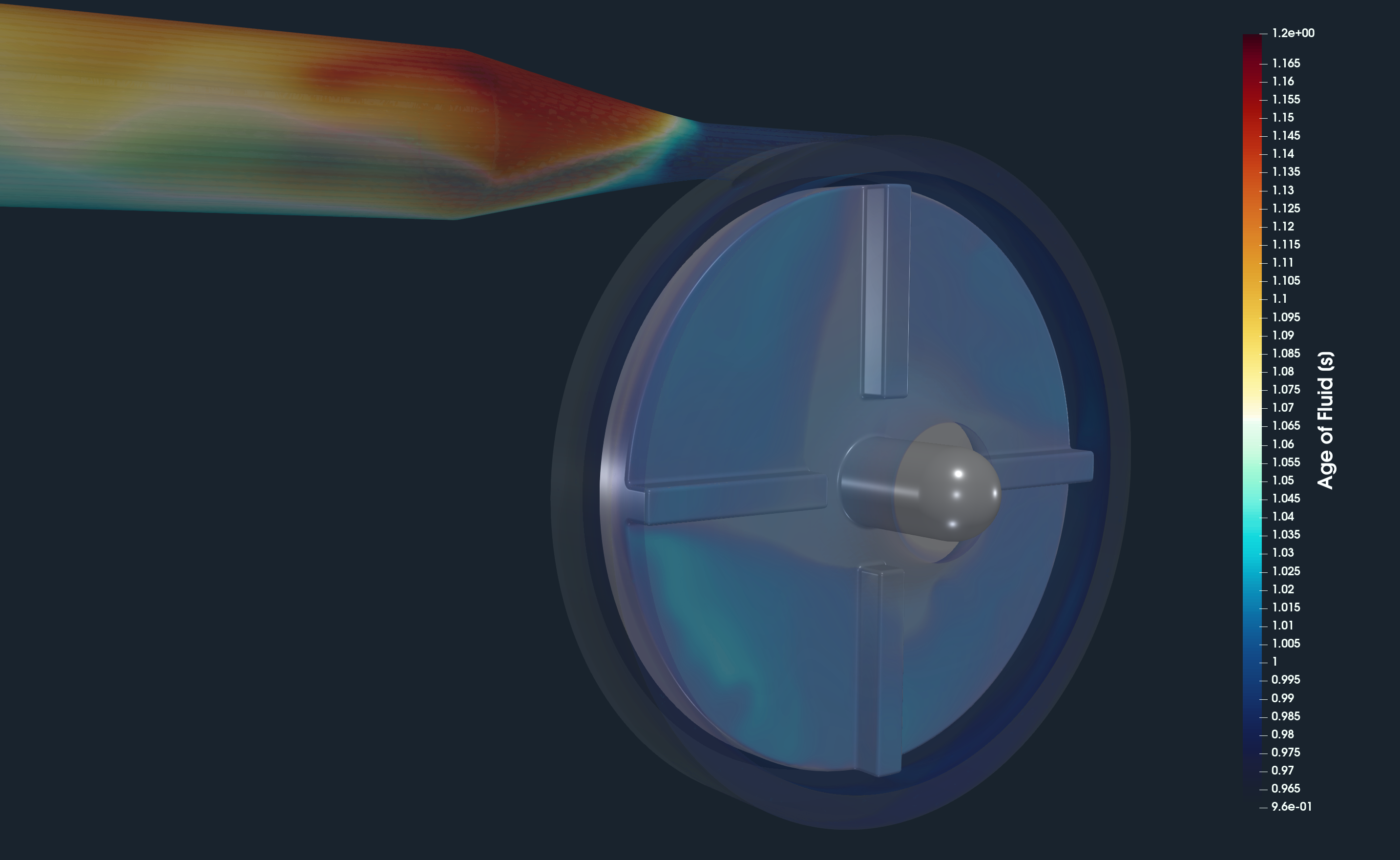
FDA Pump - Postprocessing - Age of Fluid
An important quantity is the Age the fluid. This volume field means the time [s] that the fluid spent in the flow domain since it entered it via the inlet interface. The time for which the fluid (blood) is exposed to the fluid shear stress has a significant effect on blood hemolysis. On top of that, the mean age of fluid also provides valuable information about the flow field. At rotating machinery, the locations of high age of fluid indicate flow recirculation zones which are potential areas for energy losses. In general, the age of fluid should be gradually distributed from the inlet to the outlet without sharp gradients.
FDA Pump - Postprocessing - Blood Hemolysis
Hemolysis is a process of destruction of red blood cells so that the contained oxygen-carrying pigment hemoglobin is freed into the surrounding medium. Hemolysis occurs normally in a small percentage of red blood cells as a means of removing aged cells from the bloodstream and freeing heme for iron recycling. Hemolysis is a general term for RBC damage. At pumps, the RBC get mechanically damaged by the flow shear stress. The hemolysis potential can be computed[6] and written down either as a volume field called Hemolysis Index or as a total integral (sum) over the whole computational domain.

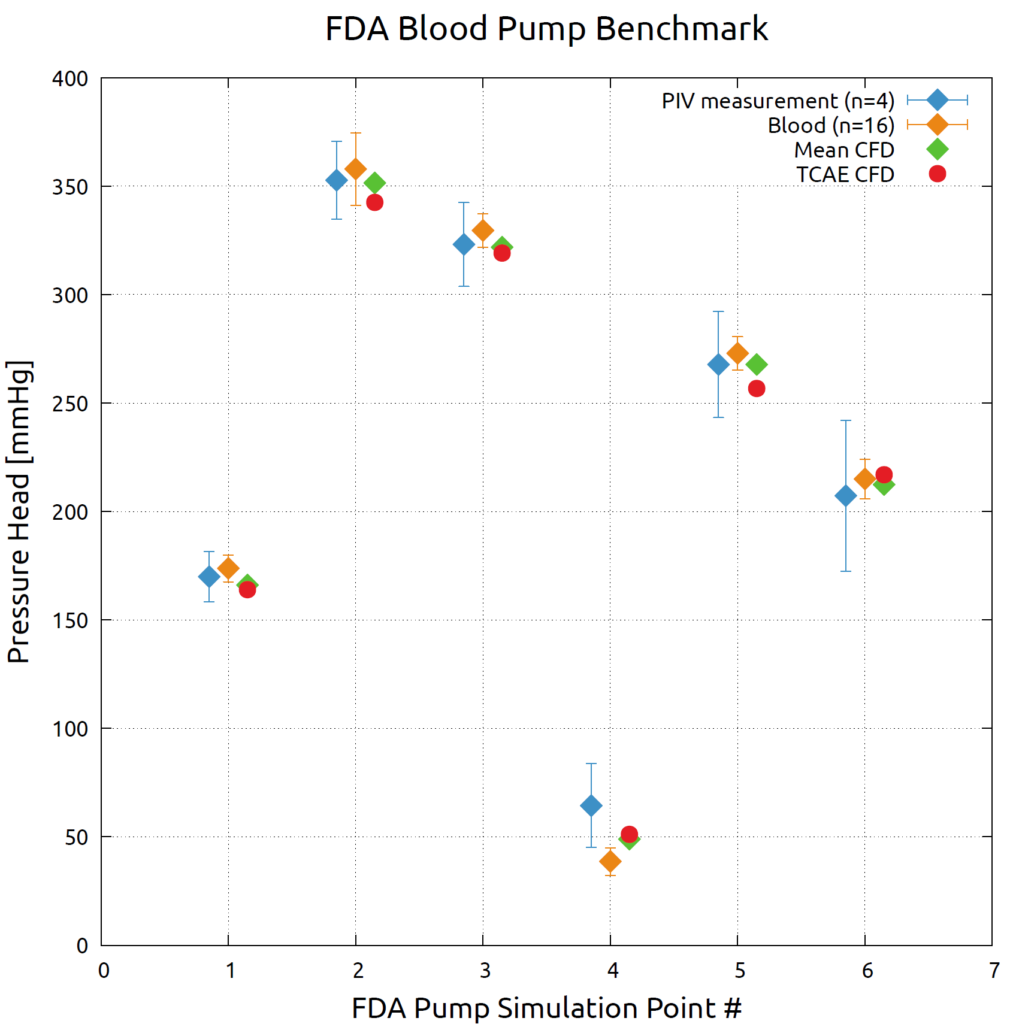
FDA Pump - Benchmark Results - Pressure Head
The FDA pump benchmark was extensively measured[1] in multiple laboratories to provide experimental velocities, pressures, and hemolysis data to support CFD validation. The measurement method was Particle Image Velocimetry (PIV). Results of PIV testing were compiled from a total of four separate data sets received from three laboratories[1]. In addition, CFD simulations were performed by more than 20 independent groups to assess available CFD techniques. The following CFD codes were used: Abaqus/CFD, AcuSolve, ANSYS CFX, ANSYS Fluent, Code_Saturne, FlowVision, SC/Tetra, STAR-CCM+, and their results we averaged in the following plot (Mean CFD)[1]. The TCAE results are plotted (red) and compared with the measurements (blue & orange) and other CFD codes (green) in the following graph. The comparison shows six simulation points for two rotation speeds (2500 & 3500 RPM) and four flow rates (2.5, 4.5, 6.0, and 7.0 l/min ) according to table A.
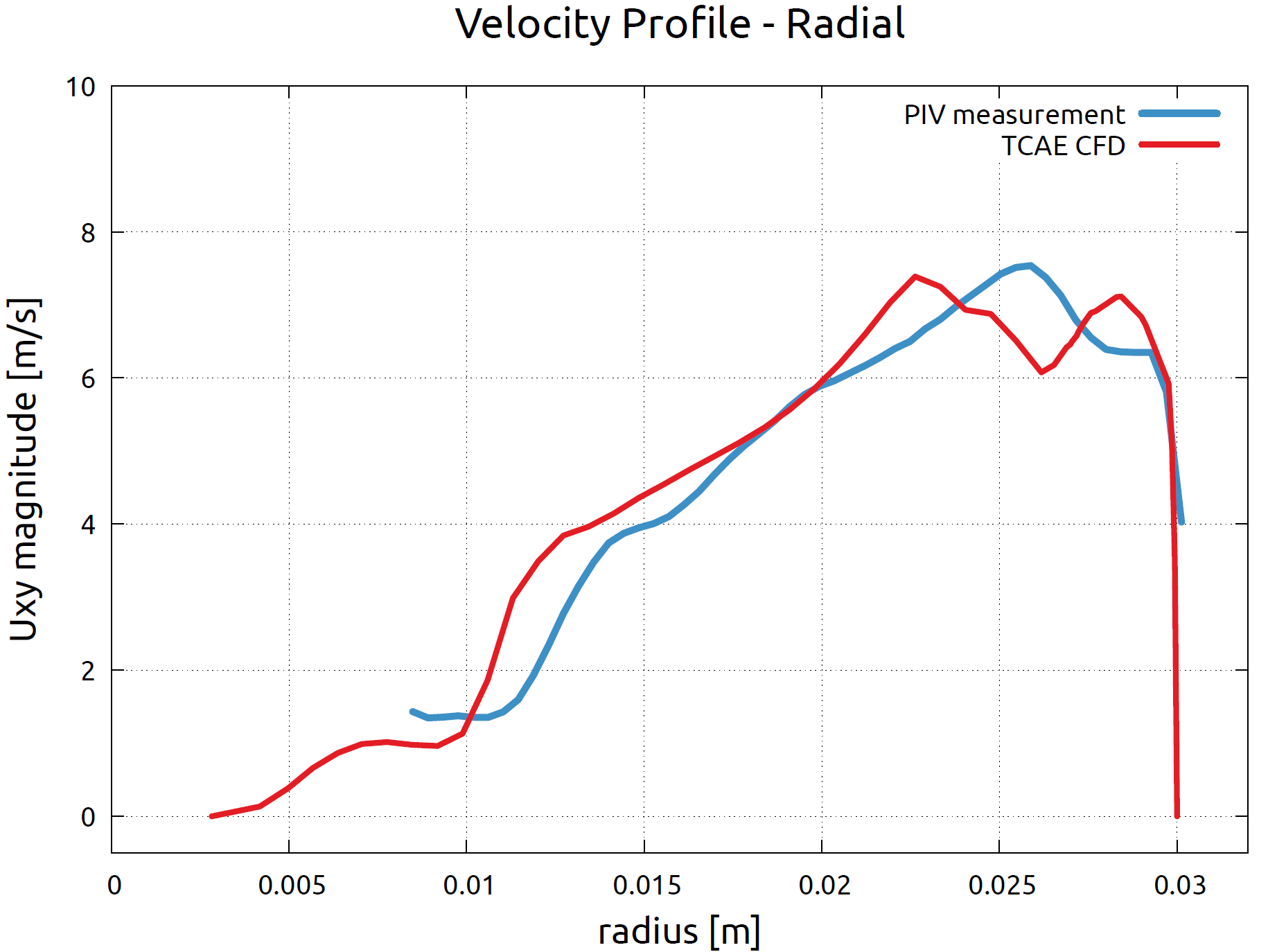
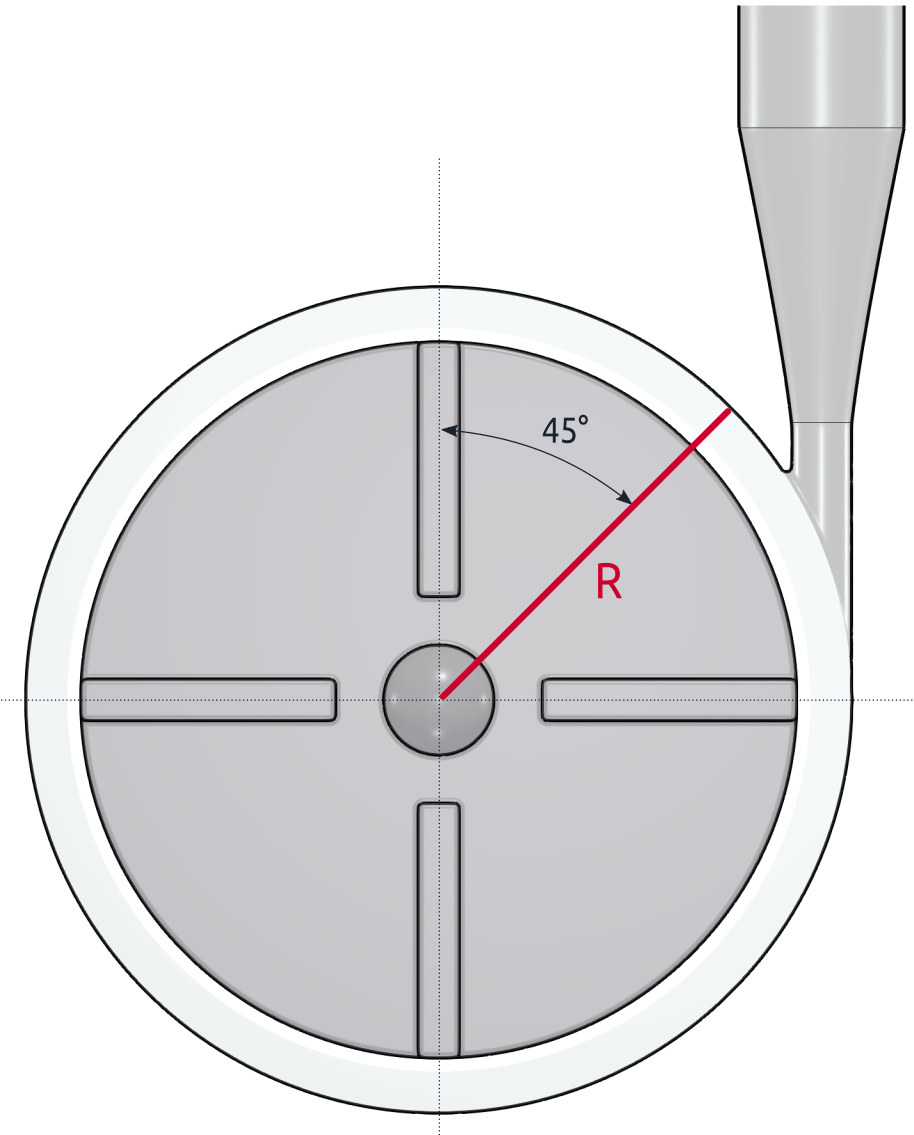
FDA Pump - Benchmark Results - Velocity Profiles
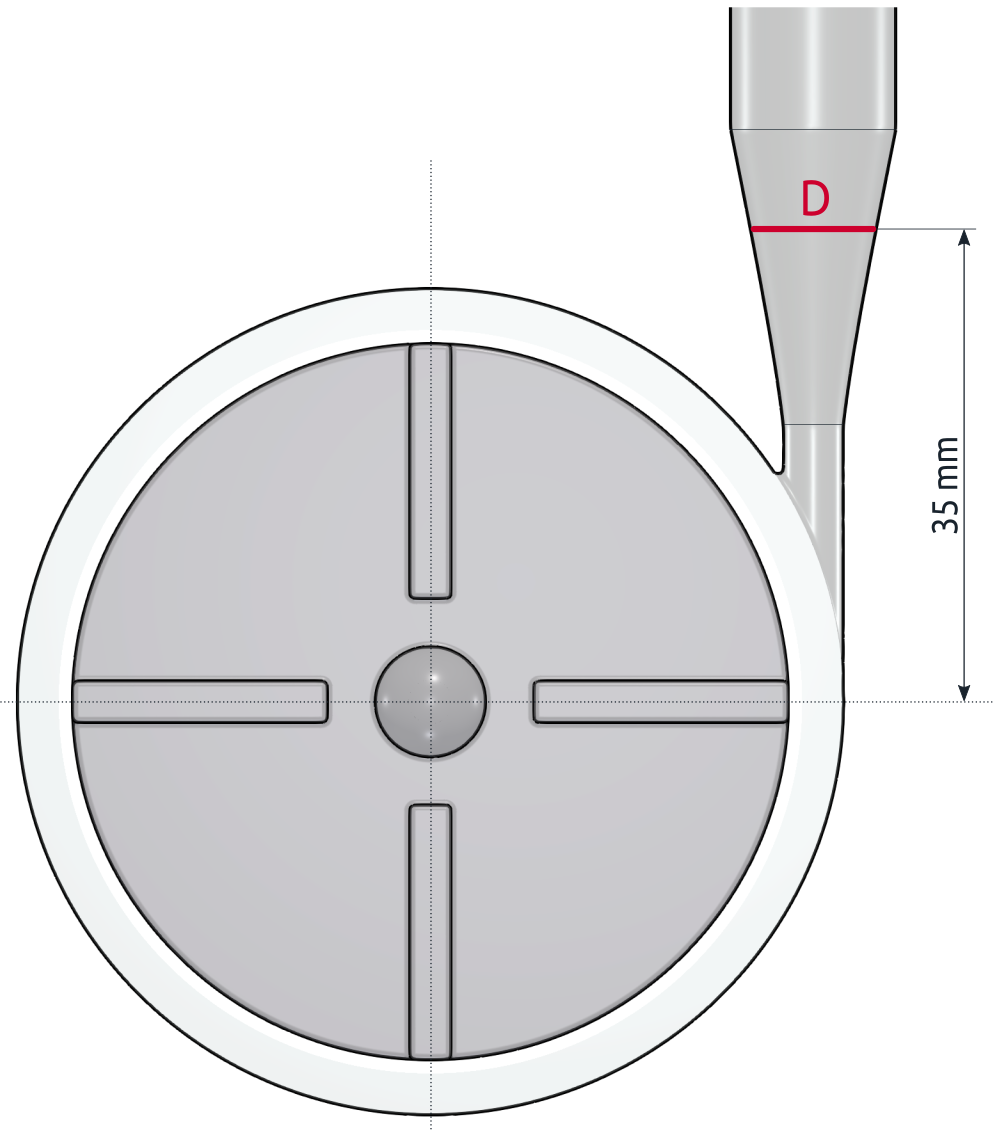
The PIV measurement has been focused on two specific locations in the flow field. The velocity profile was measured A) along the radial line R in the impeller area and B) along the mid-line D of the cross-section in the diffuser area of the pump. The images below show the comparison of the velocity profiles of the measurement and CFD simulation.
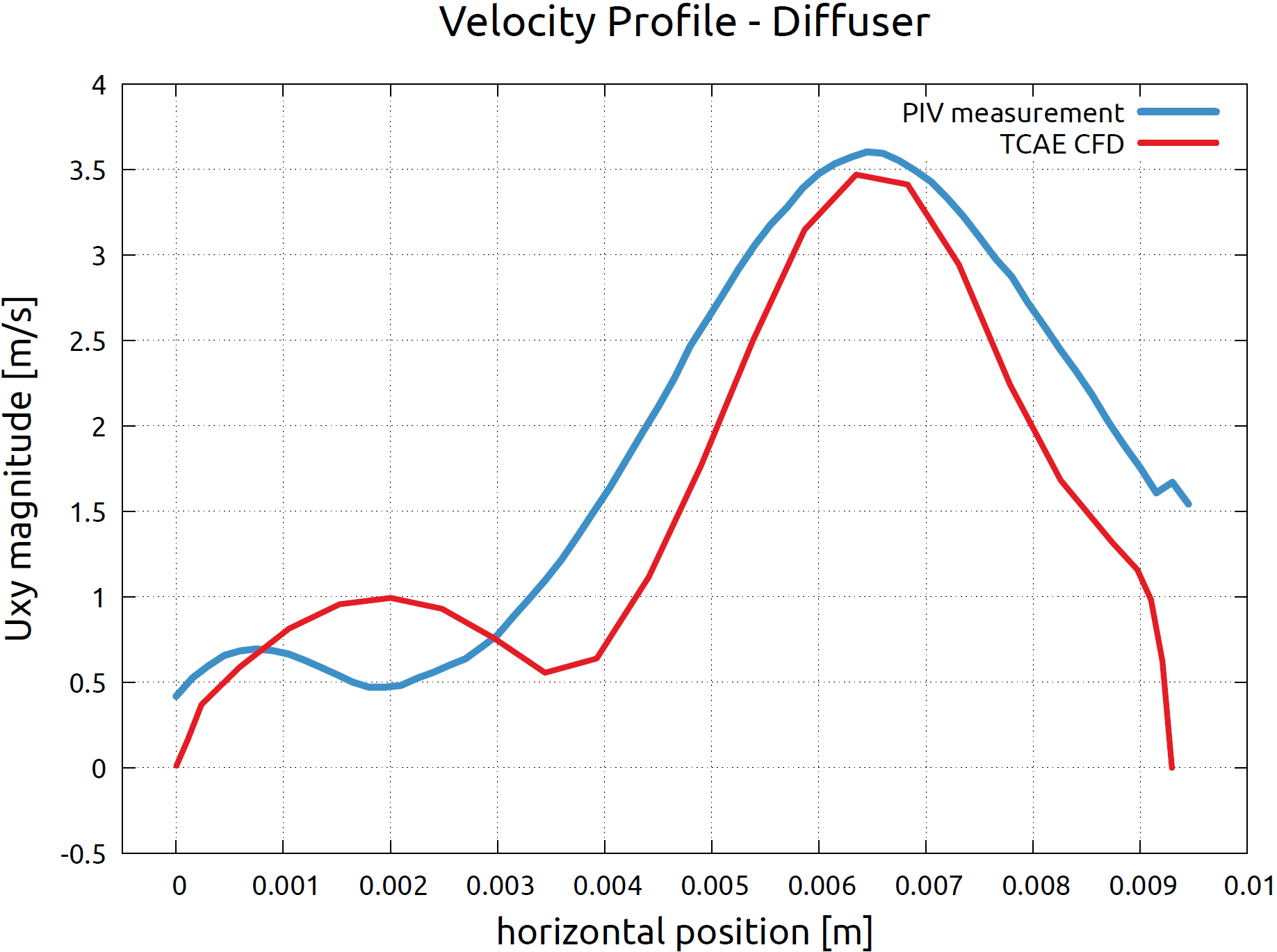
Conclusion
- It has been shown how to make a comprehensive CFD & FEA analysis including FSI of the FDA Pump in a single automated workflow.
- The TCAE results were successfully compared to the measurement data and with other CFD simulation results.
- There was no special tuning used in the CFD simulation at all. There remains a lot of space for tuning CFD methodology, especially for the mesh resolution, turbulence modeling, and numerical schemes. On the physical model level, the is enough space remaining in terms of using shear-thinning non-Newtonian models and blood viscoelasticity models.
- TCAE showed to be a very effective tool for CFD, FEA, and FSI engineering simulations for medical devices and for rotating machinery in general.
- This benchmark was intentionally written in short not to overwhelm its reader with too many details. The original intention was to show the modern simulation workflow and its accuracy.
- The benchmark details are listed in the references below[1] [2] [3] [4] [5] [6].
- All the technical details regarding CFD & FEA simulation are listed in the TCAE manual[8].
- The benchmark geometry and data are freely available for download on the CFDSUPPORT website https://www.cfdsupport.com.
- More information about TCAE can be found on CFD SUPPORT website: https://www.cfdsupport.com/tcae.html
- Questions will be happily answered via email info@cfdsupport.com.
References
[1] Malinauskas RA, Hariharan P, Day SW, Herbertson LH, Buesen M, Steinseifer U, Aycock KI, Good BC, Deutsch S, Manning KB, Craven BA. FDA Benchmark Medical Device Flow Models for CFD Validation. ASAIO J. 2017 Mar/Apr;63(2):150-160. doi: 10.1097/MAT.0000000000000499. PMID: 28114192.
[2] Good, B. C., & Manning, K. B. (2020). Computational modeling of the Food and Drug Administration’s benchmark centrifugal blood pump. Artificial organs, 44(7), E263-E276. https://doi.org/10.1111/aor.13643
[3] Heck ML, Yen A, Snyder TA, O’Rear EA, Papavassiliou DV. Flow-Field Simulations and Hemolysis Estimates for the Food and Drug Administration Critical Path Initiative Centrifugal Blood Pump. Artif Organs. 2017 Oct;41(10):E129-E140. doi: 10.1111/aor.12837. Epub 2017 Feb 7. PMID: 28168706.
[4] Karimi, M.S., Razzaghi, P., Raisee, M. et al. Stochastic simulation of the FDA centrifugal blood pump benchmark. Biomech Model Mechanobiol 20, 1871–1887 (2021). https://doi.org/10.1007/s10237-021-01482-0
[5] Nassau, CJ, Wray, TJ, & Agarwal, RK. “Computational Fluid Dynamic Analysis of a Blood Pump: An FDA Critical Path Initiative.” Proceedings of the ASME/JSME/KSME 2015 Joint Fluids Engineering Conference. Volume 2: Fora. Seoul, South Korea. July 26–31, 2015. V002T26A002. ASME.
[6] Hariharan P, D’Souza G, Horner M, Malinauskas RA, Myers MR. Verification Benchmarks to Assess the Implementation of Computational Fluid Dynamics Based Hemolysis Prediction Models. J Biomech Eng. 2015 Sep;137(9). doi: 10.1115/1.4030823. Epub 2015 Jul 9. PMID: 26065371.
[7] TCAE Training
[8] TCAE Manual
[9] TCAE Webinars
Download TCAE Tutorial - FDA Pump Benchmark
File name: FDA-pump-benchmark-TCAE-Tutorial-21.09.zip
File size: 32 MB
Tutorial Features: CFD, FEA, FSI, TCAE, TMESH, TCFD, TFEA, SIMULATION, FDA PUMP, RADIAL PUMP, CENTRIFUGAL PUMP, TURBOMACHINERY, INCOMPRESSIBLE FLOW, DEFORMATION, DISPLACEMENT, STRESS, MODAL ANALYSIS, INCOMPRESSIBLE, RANS, BLOOD FLOW, STEADY-STATE, AUTOMATION, WORKFLOW, RADIAL FLOW, FULL IMPELLER, SNAPPYHEXMESH, NETGEN, 3 COMPONENTS, RPM=3500, R=30mm
